Organic light-emitting diodes (OLEDs) have become a major player in the display industry, with a market reaching on the order of $40B in 2020 and expected to grow well over $150B by 2026. The current commercial devices, however, are based on coordination complexes involving heavy metal atoms. Thus, a lot of attention is being paid to the development of efficient purely organic emitters.
In conventional fluorescent emitters that have a closed-shell electronic structure, according to spin statistics, the recombination of the electrons and holes injected at their respective electrodes generally produces singlet and triplet excitons in a ratio of 1:3. As the triplet excitons are dark states that hardly luminesce, right from the outset, ¾ of the input electrical energy is lost. To overcome the 25% internal quantum efficiency limit imposed by spin statistics, we are currently investigating two classes of purely organic emitters.
The first class exploits the thermally activated delayed fluorescence (TADF) process. In such materials, the energy separation between the lowest singlet excited state S1 and the lowest triplet excited state T1 (ΔEST) is designed to be small (< 0.2 eV), which makes possible the up-conversion from the T1 state to the emissive S1 state via reverse intersystem crossing (RISC). The stability and color purity of TADF-based OLEDs can be further improved by making use of the hyperfluorescence approach; here, the TADF molecules are used as sensitizers that transfer their excitation energy by means of a Förster resonance energy process to fluorophores chosen for their high radiative decay rates and narrow emission spectrum.
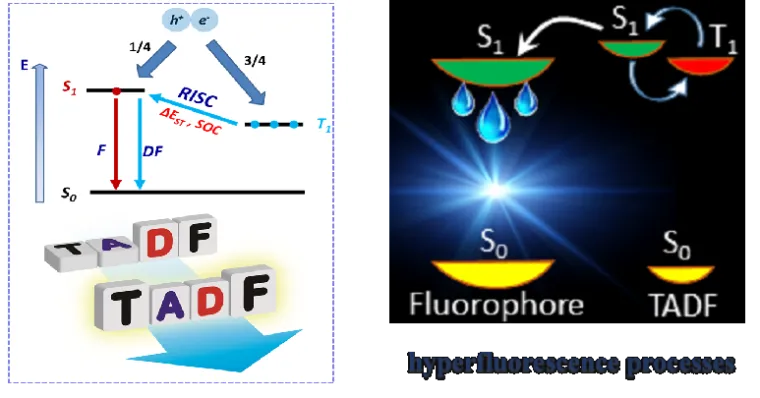
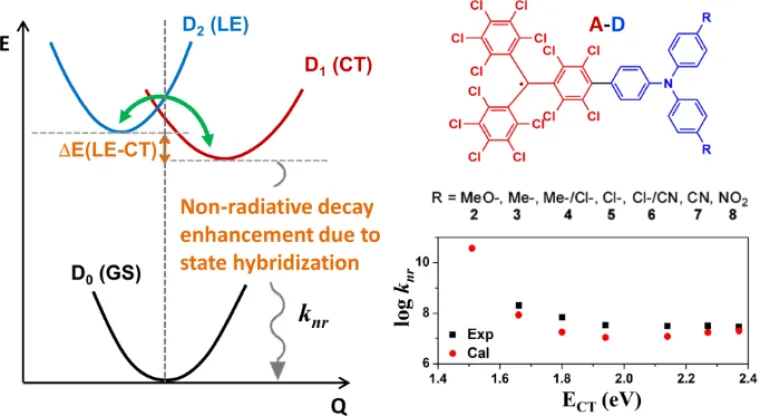
Recently, in parallel to TADF emitters, a novel design paradigm has emerged based on neutral radical-carrying organic emitters that have an open-shell electronic structure. The first excited electronic state in these radicals is a spin-doublet state (D1), which has the same spin configuration as the ground state (D0). Such an electronic structure naturally offers the way to circumvent the singlet-triplet conundrum of closed-shell emitters. In general, we use a combination of molecular dynamics simulations, quantum-chemical calculations, and theoretical modeling to investigate how the chemical structure of the emitters and the composition of the active layer affect the opto-electronic performance.
Recent publications:
-
E. Cho, L. Liu, V. Coropceanu, J. L. Brédas, “Impact of secondary donor units on the excited-state properties and thermally activated delayed fluorescence (TADF) efficiency of pentacarbazole-benzonitrile emitters”, The Journal of Chemical Physics, 153,144708 (2020).
-
H. Abroshan, Y. Zhang, X. Zhang, C. Fuentes‐Hernandez, S. Barlow, V. Coropceanu, S. R. Marder, B. Kippelen, J. L. Brédas, “Thermally Activated Delayed Fluorescence Sensitization for Highly Efficient Blue Fluorescent Emitters”, Advanced Functional Materials, 2005898 (2020).
-
H. Abroshan, V. Coropceanu, J. L. Brédas, “Radiative and Nonradiative Recombinations in Organic Radical Emitters: The Effect of Guest–Host Interactions”, Advanced Functional Materials, 30, 2002916 (2020).
-
L. S. Cui, A. J. Gillett, S. F. Zhang, H. Ye, Y. Liu, X. K. Chen, Z. S. Lin, E. W. Evans, W. K. Myers, T. K. Ronson, H. Nakanotani, S. Reineke, J. L. Bredas, C. Adachi and R. H. Friend, “Fast spin-flip enables efficient and stable organic electroluminescence from charge-transfer states”, Nature Photonics 14, 636-642, (2020).
-
H. Abroshan, V. Coropceanu and J.L. Brédas, “Hyperfluorescence-Based Emission in Purely Organic Materials: Suppression of Energy-Loss Mechanisms via Alignment of Triplet Excited States”, ACS Materials Letters 2, 1412-1418 (2020).
-
E. Cho, V. Coropceanu, and J. L. Brédas,“Organic Neutral Radical Emitters: Impact of Chemical Substitution and Electronic-State Hybridization on the Luminescence Properties”, Journal of the American Chemical Society 142, 1782-1786 (2020).
-
H. Noda, X. K. Chen, H. Nakanotani, T. Hosokai, M. Miyajima, N. Notsuka, Y. Kashima, J. L. Bredas and C. Adachi, “Critical role of intermediate electronic states for spin-flip processes in charge-transfer-type organic molecules with multiple donors and acceptors”, Nature Materials, 18, 1084-1090 (2019).
-
H. Q. Guo, Q. M. Peng, X. K. Chen, Q. Y. Gu, S. Z. Dong, E. W. Evans, A. J. Gillett, X. Ai, M. Zhang, D. Credgington, V. Coropceanu, R. H. Friend, J. L. Bredas and F. Li, “High stability and luminescence efficiency in donor-acceptor neutral radicals not following the Aufbau principle”, Nature Materials 18, 977-984 (2019).

